
Participants were randomly assigned to receive treatment with either pembrolizumab plus the chemotherapy drugs 5-fluorouracil ( 5-FU) and cisplatin, or a placebo plus the chemotherapy drugs. The study-which was funded by Merck, the manufacturer of pembrolizumab-included both people with ESCC and esophageal adenocarcinoma, which usually arises lower in the esophagus, closer to the stomach. The KEYNOTE-590 trial included about 750 people with esophageal cancer that had spread to nearby tissue or lymph nodes and couldn’t be removed with surgery. “So, when the cancer grows, approximately half of patients are typically not able to move on to a second-line therapy.”īoth of the clinical trials presented at the ESMO meeting were launched to test whether immunotherapy can improve how long patients live if given earlier in treatment. Patients tend to be sicker when we first see them compared to patients with other tumor types,” explained Harry Yoon, M.D., of the Mayo Clinic, who was not involved in either trial. “Esophageal cancer is an aggressive disease, more than some other common cancer types. 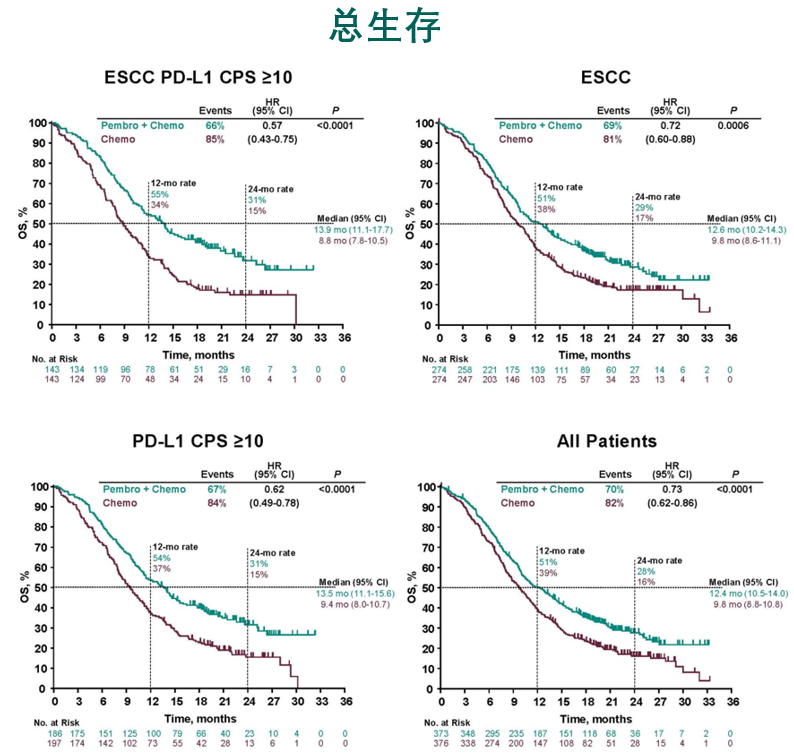
This type of esophageal cancer most often arises in the lining of the upper or middle part of the esophagus. In 2019, the Food and Drug Administration (FDA) approved the immunotherapy drug pembrolizumab (Keytruda) as second-line treatment for some people with a type of locally advanced or metastatic esophageal cancer called esophageal squamous cell carcinoma (ESCC). “Esophageal cancers have a very poor prognosis, and 5-year survival in Western countries ranges between 10% and 12%,” said Andrés Cervantes, M.D., Ph.D., an oncologist at the University of Valencia in Spain who was not involved with either study, speaking at the ESMO meeting. However, the cancer usually gets worse or spreads within a few months. So standard initial (first-line) treatment consists of systemic treatment with chemotherapy.Ĭhemotherapy can cause some advanced esophageal tumors to shrink or stop growing for a while. If the disease has spread widely, a surgeon may not be able to remove all of the cancer and surgery won’t help the patient live longer. Using Pembrolizumab Early in TreatmentĮsophageal cancer is often diagnosed at an advanced stage, when it has spread to nearby organs. But according to several experts on esophageal cancer, the results from both trials may lead to these drugs becoming new standard treatments for a cancer for which new, effective therapies have been hard to come by. The results from both studies are considered preliminary because the trials are still ongoing.  
In one of the studies, people who received a checkpoint inhibitor also lived longer overall.įindings from both trials were presented last month at the European Society for Medical Oncology (ESMO) virtual annual meeting. In both trials, treatment with drugs known as immune checkpoint inhibitors increased how long people with advanced esophageal cancer lived without their cancer getting worse, compared with those who received standard treatments. Immunotherapy may become part of early treatment for some people with advanced esophageal cancer, based on results from two large clinical trials. The approval was made based on the results of the KEYNOTE-590 trial, described in this Cancer Currents story. UPDATE: On March 22, 2021, FDA approved pembrolizumab (Keytruda) in combination with chemotherapy for some patients with esophageal or gastroesophageal cancer who are not candidates for surgery or chemotherapy and radiation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
